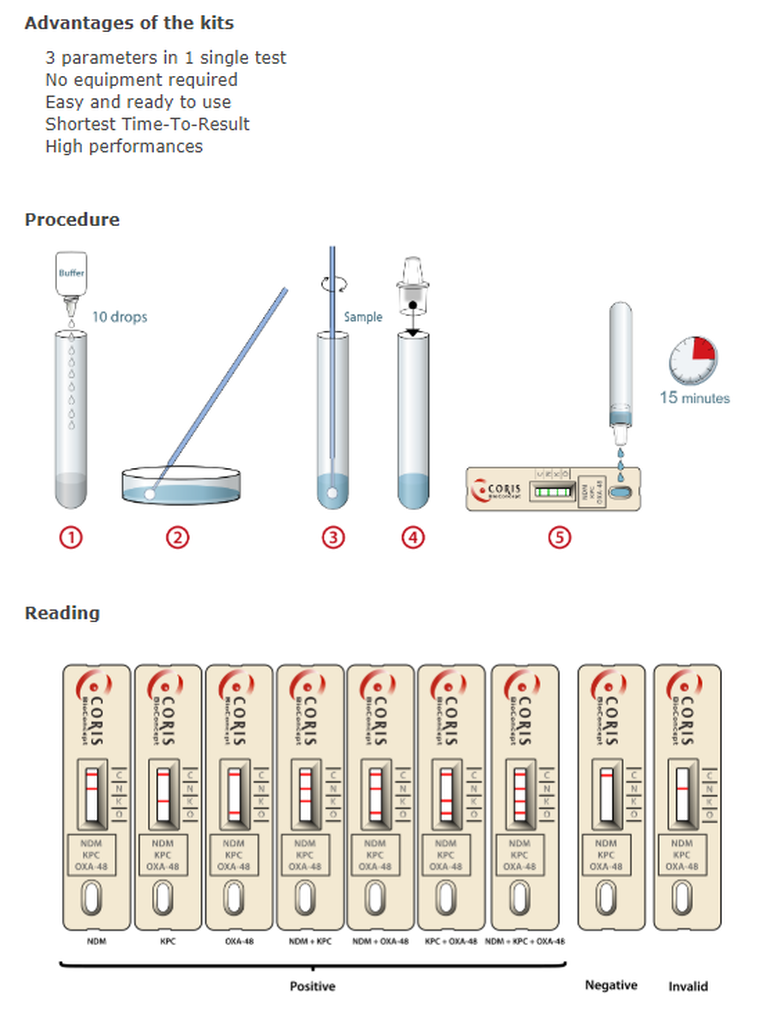
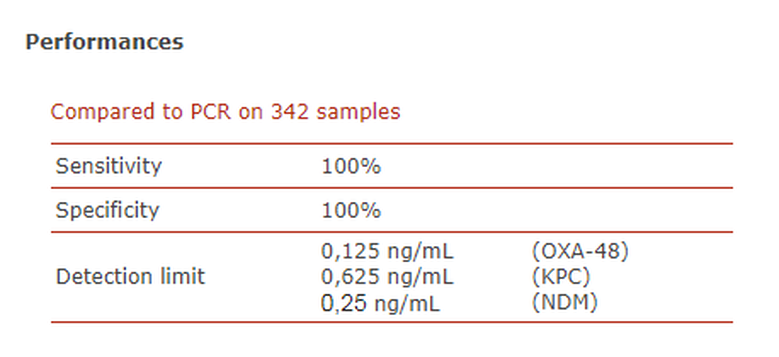
RESIST 3 O.K.N. A simple yet accurate immunochromatography test for the independent identification of OXA-48-like, KPC, and NDM carbapenemases from a bacterial colony.
RESIST-3 O.K.N. - Publications
• P2325 – Rapid detection of carbapenemase-producing organisms directly from blood cultures using a combined MALDI-TOF MS and RESIST O.K.N. workflow.
E. Wey, L. Ainsworth, T. McHugh and I. Balakrishnan.
28th European Congress of Clinical Microbiology and Infectious Diseases, Infectious Diseases April 21 – 24, 2018.
Poster
• P2297 – Retrospctive evaluation of Coris BioConcept’s RESIST-3 OKN K-SeT test to detect carbapenemase-producing organisms (CPO) from diverse genetic backgrounds.
B.M. Willey, M. Al-Ani, Y. Sokolskyy, E. Osuji, A. Paterson, DA. Boyd, W. Chui, T. Mazzulli and S.M. Poutanen.
28th European Congress of Clinical Microbiology and Infectious Diseases, Infectious Diseases April 21 – 24, 2018.
Poster
• 00812 – Prospective evaluation of Coris BioConcept’s RESIST-3 OKN K-SeT test to detect carbapenemase-producing organisms (CPO) from surveillance cultures.
B.M. Willey, M. Al-Ani, Y. Sokolskyy, E. Osuji, A. Paterson, DA. Boyd, W. Chui, T. Mazzulli and S.M. Poutanen.
28th European Congress of Clinical Microbiology and Infectious Diseases, Infectious Diseases April 21 – 24, 2018.
Poster
• E0141 – Trends in resistance mechanisms of carbapenem resistant Klebsiella pneumoniae blood isolates during a two year period in a tertiary care Hellenic hospital.
S. Tsiplakou, V. Papaioannou, E. Koiliari, D. Stefani and M. LelekisA. Saleh, S. Göttig and A. Hamprecht.
28th European Congress of Clinical Microbiology and Infectious Diseases, Infectious Diseases April 21 – 24, 2018.
Poster
• 00810 – Rapid detection of carbapenemase-producing Enterobacteriaceae directly from positive blood cultures by a new immunochromatographic assay.
A.Hamprecht, H. Seifert and A. Saleh.
28th European Congress of Clinical Microbiology and Infectious Diseases, Infectious Diseases April 21 – 24, 2018.
Poster
• Performance of “RESIST-3 O.K.N. K-SeT” immunochromatographic assay for the detection of OXA-48 like, KPC, and NDM carbapenemases in Klebsiella pneumoniae in Turkey.
P. Sağıroğlu, U. Hasdemir, G. Altınkanat Gelmez, B. Aksu, O. Kataruna and G. Söyletir.
Brazilian Journal of Microbiology 49 (2018)885–890 Available online 1 March 2018.
Abstract
In this study, the performance of the “RESIST-3 O.K.N. K-SeT” (Coris BioConcept, Gembloux, Belgium) immunochromatographic assay was evaluated in 132 Klebsiella pneumoniae comprising 102 carbapenem resistant and 30 carbapenem susceptible isolates. Genotypically known isolates of Gram negative bacteria (n = 22) including various species were also tested by the assay as controls. The isolates tested by the immunochromatographic assay and also were run PCR for blaKPC, blaIMP, blaVIM, blaNDM, and blaOXA-48. The rates of blaNDM, blaOXA-48, and blaKPC in carbapenem resistant isolates were found at 52.9%, 39.2%, and 2.0%, respectively. Both blaNDM and blaOXA-48 were found in six (5.9%) isolates. The results of the assay showed 100% concordance with those obtained by PCR in 132 K. pneumoniae. The agreement between the two methods was found to be identical at the isolate level. The assay also correctly detected all genotypically known isolates of Escherichia coli, Serratia marcescens, Citrobacter freundii, Enterobacter cloacae, K. pneumoniae carrying blaKPC, blaNDM, and/or blaOXA-48. On the other hand, the assay did not exhibit any cross-reaction in control isolates harboring blaIMP and blaVIM. We conclude that the RESIST-3 O.K.N. K-SeT is a reliable, rapid, and user friendly test and we recommend it for routine diagnostic laboratories.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6175700/pdf/main.pdf
• Multiplex immunochromatographic detection of OXA-48, KPC and NDM carbapenemases: impact of the inoculum, antibiotics and agar.
A. Saleh, S. Göttig and A. Hamprecht.
J Clin Microbiol. 2018 Feb 14. pii: JCM.00050-18.
Abstract
SYNOPSIS For rapid detection of carbapenemase-producing Enterobacteriaceae (CPE), immunochromatographic lateral flow tests (ICT) have recently been developed. The aim of this study was to assess the new multiplex ICT RESIST-3 O.K.N®. and to investigate if it can be performed directly from susceptibility testing plates. Additionally the impact of the inoculum and carbapenem disks on sensitivity and specificity was evaluated. The new ICT was challenged using 63 CRE isolates, including 51 carbapenemase producers. It was assessed using five different conditions directly from Mueller-Hinton agar (MHA): 1 μl or 10 μl inoculum harvested in the absence of antibiotic pressure or 1 μl taken from the inhibition zone of either ertapenem, imipenem or meropenem disks.The sensitivity of the ICT was 100 % for OXA-48-like and KPC and 94.4 % for NDM with the 1 μl inoculum. When harvested adjacent to a carbapenem disk, the sensitivity increased to 100 %. Additionally, with zinc-supplemented MHA both the sensitivity increased and the NDM band became visible faster (mean time 8 ± 3.9 min for MHA compared to 1.9 ± 1.5 min for MHA+zinc, P=0.0016). The specificity of the ICT was 100%.Conclusions: The RESIST-3 O.K.N. ICT is a sensitive and rapid test for the detection of three highly prevalent carbapenemases. However, false-negative results for NDM can occur. We recommend an inoculum of 1 μl that is harvested adjacent to an ertapenem or meropenem disk and the use of agars with sufficient zinc content to achieve the best performance.
http://jcm.asm.org/content/early/2018/02/09/JCM.00050-18.long
• Improving the detection of carbapenemase-producing organisms (CPO) in a low-prevalence setting: evaluation of four commercial methods and implementation of an algorithm of testing.
G. L. Vanstone, S. Woodhead, K. Roulston, H. Sharma, E. Wey, E. R. Smith, D. Mack and I. Balakrishnan.
Article is presented in J Med Microbiol. 2018 Feb;67(2):208-214.
Abstract
PURPOSE:
Carbapenemase-producing organisms (CPOs) can be resistant to almost all β-lactams and represent an increasing threat in healthcare facilities. Detection of these organisms in routine diagnostic laboratories is difficult; here we evaluate four commercially available CPO detection assays and assess their suitability for the clinical laboratory.
METHODOLOGY:
A panel of 95 clinical multidrug-resistant organisms (22 NDM, 24 OXA-48, 19 VIM, 4 OXA-23, 3 KPC, 4 NDM+OXA-48, 1 OXA23+NDM, 1 IMI, 1 IMP-1, 9 ESBL, 3 derepressed AmpC and 4 inducible AmpC producers) were tested by the RESIST-3 O.K.N., RapidEC CarbaNP, Acuitas Resistome and Xpert Carba-R assays.Results/Key Findings. The commercial assays performed well, with high sensitivities (96.2-100 %) and specificities (all, 100 %). The RapidEC CarbaNP and Acuitas Resistome were able to detect the broadest range of carbapenemase genotypes. The RESIST-3 O.K.N. and Xpert CarbaR had the shortest turnaround times, whilst the RapidEC CarbaNP was the only assay included in this study that could detect previously undescribed genotypes.
CONCLUSION:
Using an algorithm of the RapidEC CarbaNP, followed by either the RESIST-3 O.K.N. (Enterobacteriaceae) or the Xpert Carba-R (Pseudomonas aeruginosa and Acinetobacter spp.) on suspect CPOs allowed rapid in-house detection and genotyping of a high proportion of CPOs, reducing turnaround time by up to 7 days.
http://jmm.microbiologyresearch.org/content/journal/jmm/10.1099/jmm.0.000674#tab2
• OXA-48-, KPC- & NDM-type carbapenemase-producing organism detection.
G L Vanstone, E Wey, R Smith, D Mack and I Balakrishnan.
Article is presented on website of Pathology in Practice magazine and at the Institute of Biomedical Science congress, 2017 24-27 Sept in Birmingham, UK.
Abstract
Multidrug resistance exhibited by a range of microorganisms is a growing problem, a prime example being that resulting from carbapenemase production.
Here, Gemma Vanstone and colleagues assess the value of the RESIST-3 O.K.N. lateral-flow immunochromatography assay.
https://www.pathologyinpractice.com/story/22021/oxa-48-kpc-ndm-type-carbapenemase-producing-organism-detection
• Rapid Detection of Carbapenemases in Enterobacteriaceae: Evaluation of the RESIST-3 O.K.N (OXA-48, KPC, NDM) Multiplexed Lateral Flow Assay.
Wareham DW and Momin MHFA.
J. Clin. Microbiol. Accepted manuscript posted online 1 February 2017 doi:10.1128/JCM.02471-16
Abstract
The identification, treatment and control of carbapenem resistant Enterobacteriaceae (CRE) infections are a major challenge for healthcare institutions and diagnostic laboratories worldwide.…
http://jcm.asm.org/content/early/2017/01/27/JCM.02471-16.full.pdf+html?sid=43d81657-15d6-4722-9787-9de8e6b7c783
• Rapid Detection of OXA-48, KPC and NDM-type carbapenemase producing organisms (CPOs) using the RESIST-3 O.K.N. assay.
G. L. Vanstone, E. Wey, R. Smith, D. Mack, I. Balakrishnan
Poster
• Prospective evaluation of the OKN K-SeT assay, a new multiplex immunochromatographic test for the rapid detection of OXA-48-like, KPC and NDM carbapenemases.
Glupczynski Y, Jousset A, Evrard S, Bonnin RA, Huang TD, Dortet L, Bogaerts P, Naas T.
J Antimicrob Chemother. 2017 Mar 22
Abstract
Objectives:
There is an urgent need for accurate and fast diagnostic tests capable of identifying carbapenemase producers. Here, we assessed the performance of a new multiplex lateral flow assay (OKN K -SeT) for the rapid detection of OXA-48-like, KPC and NDM carbapenemase-producing Enterobacteriaceae from culture colonies.
Methods:
Two hundred collection isolates with characterized β-lactamase content and 183 non-duplicate consecutive isolates referred to two National Reference Centres over a 2 month period in 2016 were used to evaluate the OKN K -SeT assay.
Results:
The assay correctly detected all 42 OXA-48-like-, 27 KPC- and 30 NDM-producing isolates from the collection panel, including 7 isolates that co-produced NDM and OXA-181 carbapenemases. No cross-reactivity was observed with non-targeted carbapenemases ( n = 41) or with non-carbapenemase producers ( n = 60). Prospectively, all OXA-48-like ( n = 69), KPC ( n = 9) and NDM ( n = 19) carbapenemase-producing Enterobacteriaceae isolates were correctly detected, while 11 carbapenemase producers not targeted by the assay went undetected [VIM ( n = 8) and OXA-23/OXA-58-like ( n = 3)]. Overall, the sensitivity and specificity of the assay were 100%.
Conclusions:
The OKN assay is efficient, rapid and easy to implement in the workflow of a clinical microbiology laboratory for the confirmation of OXA-48, NDM and KPC carbapenemases. This test represents a powerful diagnostic tool as it enables the rapid detection of the most clinically important carbapenemases without the need for more costly and less frequently available molecular assays.
https://www.ncbi.nlm.nih.gov/pubmed/28369469
• P2325 – Rapid detection of carbapenemase-producing organisms directly from blood cultures using a combined MALDI-TOF MS and RESIST O.K.N. workflow.
E. Wey, L. Ainsworth, T. McHugh and I. Balakrishnan.
28th European Congress of Clinical Microbiology and Infectious Diseases, Infectious Diseases April 21 – 24, 2018.
Poster
• P2297 – Retrospctive evaluation of Coris BioConcept’s RESIST-3 OKN K-SeT test to detect carbapenemase-producing organisms (CPO) from diverse genetic backgrounds.
B.M. Willey, M. Al-Ani, Y. Sokolskyy, E. Osuji, A. Paterson, DA. Boyd, W. Chui, T. Mazzulli and S.M. Poutanen.
28th European Congress of Clinical Microbiology and Infectious Diseases, Infectious Diseases April 21 – 24, 2018.
Poster
• 00812 – Prospective evaluation of Coris BioConcept’s RESIST-3 OKN K-SeT test to detect carbapenemase-producing organisms (CPO) from surveillance cultures.
B.M. Willey, M. Al-Ani, Y. Sokolskyy, E. Osuji, A. Paterson, DA. Boyd, W. Chui, T. Mazzulli and S.M. Poutanen.
28th European Congress of Clinical Microbiology and Infectious Diseases, Infectious Diseases April 21 – 24, 2018.
Poster
• E0141 – Trends in resistance mechanisms of carbapenem resistant Klebsiella pneumoniae blood isolates during a two year period in a tertiary care Hellenic hospital.
S. Tsiplakou, V. Papaioannou, E. Koiliari, D. Stefani and M. LelekisA. Saleh, S. Göttig and A. Hamprecht.
28th European Congress of Clinical Microbiology and Infectious Diseases, Infectious Diseases April 21 – 24, 2018.
Poster
• 00810 – Rapid detection of carbapenemase-producing Enterobacteriaceae directly from positive blood cultures by a new immunochromatographic assay.
A.Hamprecht, H. Seifert and A. Saleh.
28th European Congress of Clinical Microbiology and Infectious Diseases, Infectious Diseases April 21 – 24, 2018.
Poster
• Performance of “RESIST-3 O.K.N. K-SeT” immunochromatographic assay for the detection of OXA-48 like, KPC, and NDM carbapenemases in Klebsiella pneumoniae in Turkey.
P. Sağıroğlu, U. Hasdemir, G. Altınkanat Gelmez, B. Aksu, O. Kataruna and G. Söyletir.
Brazilian Journal of Microbiology 49 (2018)885–890 Available online 1 March 2018.
Abstract
In this study, the performance of the “RESIST-3 O.K.N. K-SeT” (Coris BioConcept, Gembloux, Belgium) immunochromatographic assay was evaluated in 132 Klebsiella pneumoniae comprising 102 carbapenem resistant and 30 carbapenem susceptible isolates. Genotypically known isolates of Gram negative bacteria (n = 22) including various species were also tested by the assay as controls. The isolates tested by the immunochromatographic assay and also were run PCR for blaKPC, blaIMP, blaVIM, blaNDM, and blaOXA-48. The rates of blaNDM, blaOXA-48, and blaKPC in carbapenem resistant isolates were found at 52.9%, 39.2%, and 2.0%, respectively. Both blaNDM and blaOXA-48 were found in six (5.9%) isolates. The results of the assay showed 100% concordance with those obtained by PCR in 132 K. pneumoniae. The agreement between the two methods was found to be identical at the isolate level. The assay also correctly detected all genotypically known isolates of Escherichia coli, Serratia marcescens, Citrobacter freundii, Enterobacter cloacae, K. pneumoniae carrying blaKPC, blaNDM, and/or blaOXA-48. On the other hand, the assay did not exhibit any cross-reaction in control isolates harboring blaIMP and blaVIM. We conclude that the RESIST-3 O.K.N. K-SeT is a reliable, rapid, and user friendly test and we recommend it for routine diagnostic laboratories.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6175700/pdf/main.pdf
• Multiplex immunochromatographic detection of OXA-48, KPC and NDM carbapenemases: impact of the inoculum, antibiotics and agar.
A. Saleh, S. Göttig and A. Hamprecht.
J Clin Microbiol. 2018 Feb 14. pii: JCM.00050-18.
Abstract
SYNOPSIS For rapid detection of carbapenemase-producing Enterobacteriaceae (CPE), immunochromatographic lateral flow tests (ICT) have recently been developed. The aim of this study was to assess the new multiplex ICT RESIST-3 O.K.N®. and to investigate if it can be performed directly from susceptibility testing plates. Additionally the impact of the inoculum and carbapenem disks on sensitivity and specificity was evaluated. The new ICT was challenged using 63 CRE isolates, including 51 carbapenemase producers. It was assessed using five different conditions directly from Mueller-Hinton agar (MHA): 1 μl or 10 μl inoculum harvested in the absence of antibiotic pressure or 1 μl taken from the inhibition zone of either ertapenem, imipenem or meropenem disks.The sensitivity of the ICT was 100 % for OXA-48-like and KPC and 94.4 % for NDM with the 1 μl inoculum. When harvested adjacent to a carbapenem disk, the sensitivity increased to 100 %. Additionally, with zinc-supplemented MHA both the sensitivity increased and the NDM band became visible faster (mean time 8 ± 3.9 min for MHA compared to 1.9 ± 1.5 min for MHA+zinc, P=0.0016). The specificity of the ICT was 100%.Conclusions: The RESIST-3 O.K.N. ICT is a sensitive and rapid test for the detection of three highly prevalent carbapenemases. However, false-negative results for NDM can occur. We recommend an inoculum of 1 μl that is harvested adjacent to an ertapenem or meropenem disk and the use of agars with sufficient zinc content to achieve the best performance.
http://jcm.asm.org/content/early/2018/02/09/JCM.00050-18.long
• Improving the detection of carbapenemase-producing organisms (CPO) in a low-prevalence setting: evaluation of four commercial methods and implementation of an algorithm of testing.
G. L. Vanstone, S. Woodhead, K. Roulston, H. Sharma, E. Wey, E. R. Smith, D. Mack and I. Balakrishnan.
Article is presented in J Med Microbiol. 2018 Feb;67(2):208-214.
Abstract
PURPOSE:
Carbapenemase-producing organisms (CPOs) can be resistant to almost all β-lactams and represent an increasing threat in healthcare facilities. Detection of these organisms in routine diagnostic laboratories is difficult; here we evaluate four commercially available CPO detection assays and assess their suitability for the clinical laboratory.
METHODOLOGY:
A panel of 95 clinical multidrug-resistant organisms (22 NDM, 24 OXA-48, 19 VIM, 4 OXA-23, 3 KPC, 4 NDM+OXA-48, 1 OXA23+NDM, 1 IMI, 1 IMP-1, 9 ESBL, 3 derepressed AmpC and 4 inducible AmpC producers) were tested by the RESIST-3 O.K.N., RapidEC CarbaNP, Acuitas Resistome and Xpert Carba-R assays.Results/Key Findings. The commercial assays performed well, with high sensitivities (96.2-100 %) and specificities (all, 100 %). The RapidEC CarbaNP and Acuitas Resistome were able to detect the broadest range of carbapenemase genotypes. The RESIST-3 O.K.N. and Xpert CarbaR had the shortest turnaround times, whilst the RapidEC CarbaNP was the only assay included in this study that could detect previously undescribed genotypes.
CONCLUSION:
Using an algorithm of the RapidEC CarbaNP, followed by either the RESIST-3 O.K.N. (Enterobacteriaceae) or the Xpert Carba-R (Pseudomonas aeruginosa and Acinetobacter spp.) on suspect CPOs allowed rapid in-house detection and genotyping of a high proportion of CPOs, reducing turnaround time by up to 7 days.
http://jmm.microbiologyresearch.org/content/journal/jmm/10.1099/jmm.0.000674#tab2
• OXA-48-, KPC- & NDM-type carbapenemase-producing organism detection.
G L Vanstone, E Wey, R Smith, D Mack and I Balakrishnan.
Article is presented on website of Pathology in Practice magazine and at the Institute of Biomedical Science congress, 2017 24-27 Sept in Birmingham, UK.
Abstract
Multidrug resistance exhibited by a range of microorganisms is a growing problem, a prime example being that resulting from carbapenemase production.
Here, Gemma Vanstone and colleagues assess the value of the RESIST-3 O.K.N. lateral-flow immunochromatography assay.
https://www.pathologyinpractice.com/story/22021/oxa-48-kpc-ndm-type-carbapenemase-producing-organism-detection
• Rapid Detection of Carbapenemases in Enterobacteriaceae: Evaluation of the RESIST-3 O.K.N (OXA-48, KPC, NDM) Multiplexed Lateral Flow Assay.
Wareham DW and Momin MHFA.
J. Clin. Microbiol. Accepted manuscript posted online 1 February 2017 doi:10.1128/JCM.02471-16
Abstract
The identification, treatment and control of carbapenem resistant Enterobacteriaceae (CRE) infections are a major challenge for healthcare institutions and diagnostic laboratories worldwide.…
http://jcm.asm.org/content/early/2017/01/27/JCM.02471-16.full.pdf+html?sid=43d81657-15d6-4722-9787-9de8e6b7c783
• Rapid Detection of OXA-48, KPC and NDM-type carbapenemase producing organisms (CPOs) using the RESIST-3 O.K.N. assay.
G. L. Vanstone, E. Wey, R. Smith, D. Mack, I. Balakrishnan
Poster
• Prospective evaluation of the OKN K-SeT assay, a new multiplex immunochromatographic test for the rapid detection of OXA-48-like, KPC and NDM carbapenemases.
Glupczynski Y, Jousset A, Evrard S, Bonnin RA, Huang TD, Dortet L, Bogaerts P, Naas T.
J Antimicrob Chemother. 2017 Mar 22
Abstract
Objectives:
There is an urgent need for accurate and fast diagnostic tests capable of identifying carbapenemase producers. Here, we assessed the performance of a new multiplex lateral flow assay (OKN K -SeT) for the rapid detection of OXA-48-like, KPC and NDM carbapenemase-producing Enterobacteriaceae from culture colonies.
Methods:
Two hundred collection isolates with characterized β-lactamase content and 183 non-duplicate consecutive isolates referred to two National Reference Centres over a 2 month period in 2016 were used to evaluate the OKN K -SeT assay.
Results:
The assay correctly detected all 42 OXA-48-like-, 27 KPC- and 30 NDM-producing isolates from the collection panel, including 7 isolates that co-produced NDM and OXA-181 carbapenemases. No cross-reactivity was observed with non-targeted carbapenemases ( n = 41) or with non-carbapenemase producers ( n = 60). Prospectively, all OXA-48-like ( n = 69), KPC ( n = 9) and NDM ( n = 19) carbapenemase-producing Enterobacteriaceae isolates were correctly detected, while 11 carbapenemase producers not targeted by the assay went undetected [VIM ( n = 8) and OXA-23/OXA-58-like ( n = 3)]. Overall, the sensitivity and specificity of the assay were 100%.
Conclusions:
The OKN assay is efficient, rapid and easy to implement in the workflow of a clinical microbiology laboratory for the confirmation of OXA-48, NDM and KPC carbapenemases. This test represents a powerful diagnostic tool as it enables the rapid detection of the most clinically important carbapenemases without the need for more costly and less frequently available molecular assays.
https://www.ncbi.nlm.nih.gov/pubmed/28369469